FlyBand Exo-Skeleton
The Unmet Need
- –25% of patients with diabetes will experience Diabetic Foot Ulcers. Once treated, most patient re-ulcerate within two years, leading to progressive loss and amputation
- –Diabetic Foot Ulcer costs more than lung cancer and has a higher 3 year mortality than five of the top 10 cancers
- –Lower limb injuries such as ankle sprain, Achilles tendonopathies and plantar fasciitis are the
The Innovation
- –Results Group has developed novel exo-skeletons that integrate within footwear and clothing that can re-direct forces outside of the body – preventing foot ulcers by making it easier to walk, and also easier to heal from a variety of foot and ankle conditions
- –Robotics
Research

–FlyBand® exo-skeleton systems reduce >20% load on patients with Diabetic Foot Ulcer. (Funded by NIDILRR SBIR Phase I) Collaboration with University of Arizona, the Southwest Limb Salvage Alliance, and Baylor College of Medicine.

–FlyBand® exo-skeleton footwear systems improve launch velocity when jumping off force platform and maintain postural stability (NIH R-24 Proof of Principle Testing) Collaboration with University of Connecticut, University of Virginia.
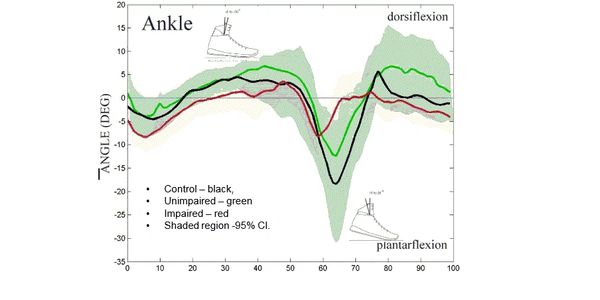
–Article in-press – Journal of Diabetes Science and Technology,
Bootstrap
This video illustrates the Bootstrap concept. Once referred to as Achilles during the early development.
Tekscan- Subject 024
Gait clinic study subject 024- Diabetic footwear. Bootstrap Phase 1
Commercialization
–Three patents issued
–Results Group will eventually spin-out a new organization to manufacture FlyBand footwear for specialized medical and industrial applications.
–Robotic Foundations will follow
Preventing Blindness
The Unmet Need
–Although remarkable treatments are widely available, eye diseases such as Macular Degeneration and Diabetic Retinopathies continue to be the leading cause of adult blindness
–The biggest unmet need is not a lack of treatment, it is the lack of ability for patients to start that treatment early enough to make a difference
–Our studies showed patients often take 3 to 5 months between the onset of disease and start of treatment, losing vision which cannot be restored
The Innovation
–Results Group has developed novel vision tests that allow patients to easily monitor their vision at home, improve confidence in their visual health, and seek treatment earlier than the standard of care
Photo Gallery
RESEARCH
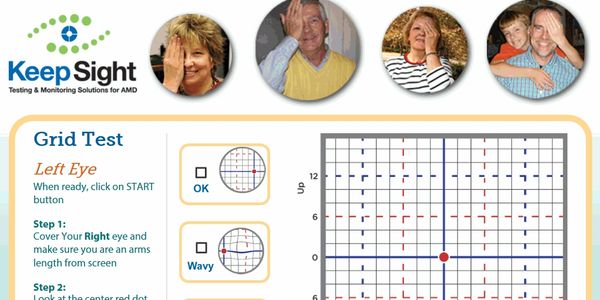
Developed Tests
Home vision tests developed by Results Group show improvement of over 500% (Funded by National Eye Institute, NIH) Collaboration with Johns Hopkins University, Wilmer Eye Institute, Nova University.
Publication:

Engaging Patients to Prevent Delay
Patients lose an average of >4 lines of vision before seeing their primary care eye doctor and being referred to a specialist (Funded internally by RG)
The largest reason for patient delay in seeking help is attributed to lack of self-efficacy (Funded internally by RG)
Studies using KeepSight abroad
Use of home vision testing in patients with AMD after initial treatment. (Funded by National Health Services, UK) Collaboration with Kings College, Belfast. Underway.
Copyright © 2011-2019 Results Group, LLC - All Rights Reserved.